Vacuna AstraZeneca no contiene tejido pulmonar de un feto abortado
En un video alertan que este producto contiene “células de fetos” que serán incrustadas en las campañas de vacunación contra la COVID-19
En un video alertan que este producto contiene “células de fetos” que serán incrustadas en las campañas de vacunación contra la COVID-19

“Esto es un hecho, es el empaquetado de AstraZeneca”, inicia la voz en off de un video que circula esta semana en las redes sociales, donde muestra una supuesta caja de la farmacéutica, que es una de las que está en la carrera por terminar la vacuna contra la COVID-19, alertando que en sus especificaciones dice que se usó tejido pulmonar de varón abortado de catorce meses. El contenido de este video es falso.
Este video circula en cadena por Facebook y WhatsApp, donde se alerta de los peligros de la vacuna en contra de la COVID-19, de la farmacéutica AstraZeneca, indicando que en la misma caja están las especificaciones que citan que se utilizó la línea celular MRC-5 humano.
“Y mi mensaje es que esto está justo delante de vuestra cara, que esto está en la vacuna”, alerta la voz con tono español, mientras muestra el significado de recombinación de ADN en Wikipedia.
Dice que la mayoría de las personas al ver términos científicos “complicados”, salta esa parte de las especificaciones. “¡No, vamos a buscarlo!”, aconseja.
Muestra que al buscar el resultado de qué significa MRC-5 se encuentra que es originalmente desarrollado a partir del tejido pulmonar del feto de un varón caucásico abortado de catorce semanas. “Ahí lo tienes. Esta vacuna COVID-19 que todos dicen que va a salvar al mundo, contiene tejido pulmonar de un feto abortado”.
Según el video, que está traducido en diferentes idiomas, esta vacuna enfermará y matará a millones de personas en el mundo.
Vacunas no contienen tejidos de células de fetos abortados
En el video se aconseja acudir al repositorio de “pre-publicaciones” científicas Research Square, donde encuentra supuestamente un texto en inglés de la vacuna ChAdOx1 que es la que está realizando la farmacéutica AstraZeneca con la Universidad de Oxford.
Research Square, es una plataforma que tiene documentos de ciencia que no han sido validados por científicos independientes al estudio.
“Hago este vídeo para que aprendan a indagar por ustedes mismos”, dice la persona que muestra las imágenes en una pantalla.
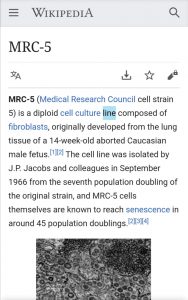
Al encontrar la supuesta caja de esta vacuna, se lee “MRC-5”, cuyo significado es buscado en Wikipedia en este video.
Al buscar sobre MRC-5 en Wikipedia, efectivamente dice que es una línea de cultivo de células desarrollado originalmente a partir del “tejido pulmonar de un feto varón caucásico abortado de catorce semanas”.
Sin embargo esa publicación de Wikipedia agrega que la línea celular fue aislada por JP Jacobs y sus colegas en septiembre de 1966 a partir de la séptima “duplicación” de la población de la cepa original.
“Se sabe que las células MRC-5 mismas alcanzan la senescencia en alrededor de 45 duplicaciones de población”, indica este portal web.
A partir de las células de esa línea, se han producido vacunas para la triple vírica, para la varicela y para la polio.
Es decir, esas células han sido duplicadas para diferentes tipos de vacunas, no fueron sacadas de fetos.
¿Y el feto?
Según el sitio web Historia de las vacunas, estas células fueron encontradas tras un aborto terapéutico, donde se descubrió que el feto tenía alguna enfermedad grave.
No fue un aborto realizado para investigar el feto, fue el análisis posterior que dio como resultado la extracción de tejidos, de donde resultaron las líneas celulares.
No se han utilizado nuevos tejidos para mantener esta línea desde entonces, es decir desde 1966.
El sitio web de “History of Vaccines” es miembro del proyecto Vaccine Safety Net (VSN) dirigido por la Organización Mundial de la Salud (OMS).
Una verificación anterior de Maldita.es asegura que ninguna vacuna contiene células de fetos abortados.
“Para generar esos virus hay que cultivarlos, y eso no puede hacerse en cualquier superficie”, explica.
Entonces, los virus necesitan células a las que infectar para sobrevivir, y la opción más eficiente es el uso de cultivos celulares, “células que se cultivan en un medio artificial de forma controlada”.
Estado de la vacuna
La empresa británica AstraZeneca resalta en su portal web que la vacuna “AZD1222” alcanzó el criterio de “valoración principal de eficacia” en la prevención de COVID-19, según informe del 23 de noviembre.
Este informe refiere que los resultados “positivos de alto nivel” de un análisis intermedio de ensayos clínicos de AZD1222 en el Reino Unido y Brasil, mostraron que la vacuna fue “muy eficaz” para prevenir la COVID-19.
El criterio de valoración principal, es que “no se informaron hospitalizaciones ni casos graves de la enfermedad en los participantes que recibieron el tratamiento”, indica su informe.
Lo que viene, según la farmacéutica es preparar “inmediatamente” la presentación regulatoria de los datos a las autoridades de todo el mundo “que tienen un marco establecido” para la aprobación condicional o anticipada.





